Breaking Through
Advancing medicine by way of research
Written by Sonia Fernandez '03 & Shelly Leachman
Photos by Matt Perko
Harnessing their curiosity and ingenuity, researchers in disciplines from biology and chemistry to engineering, psychology, and the humanities, are bending their efforts toward improving outcomes for doctors and making life better for patients. They’re pioneering new diagnostic approaches and tests, innovating new therapies for diseases and chronic conditions, and advancing our understanding of life’s most confounding medical challenges. They’re leveraging their technical and technological prowess, sharing their scientific expertise and, importantly, passing their knowledge on to the next generation of researchers. Here we highlight six scientists — a modest sampling from the ever-growing number of passionate medical changemakers across campus — who are making inroads on conditions that affect millions of people around the world.

The work
Small but mighty macrophages, part of the innate immune system, are big eaters. These white blood cells surround and kill microorganisms and remove dead cells in a process called phagocytosis. When they detect a threat, macrophages can also spark action in other immune cells. What if they could be programmed to identify and eat cancer? That’s what Meghan Morrissey and her lab are working toward, aiming to advance immunotherapy for cancerous tumors. Employing a strategy analogous to one that proved revolutionary for T cells — and was FDA approved as a therapy — Morrissey’s team hopes to train macrophages to eat cancer by swapping one part of a cellular protein with antibody fragments that recognize cancer cells. The approach could one day provide an alternative to T cell therapies, which comprise the majority of currently available immunotherapies.
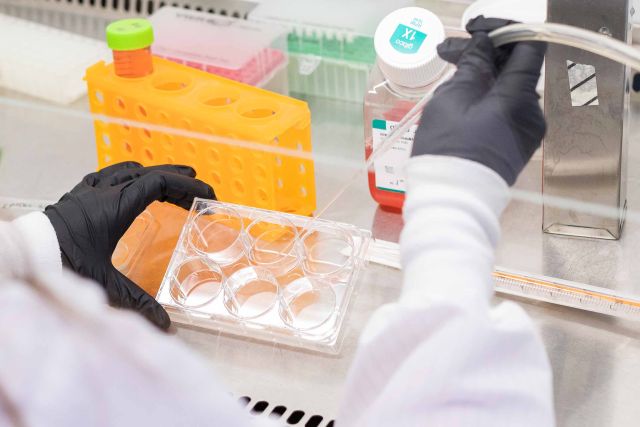
The why
“It’s a terrible disease. Everybody knows someone who is affected by cancer. I cannot overstate that as a motivation. And every cancer is unique — each comes from a different set of mutations — so the scope of the problem is big and complex. But out of this incredible mess of millions of people being affected by slightly different cancers, we have to find some sort of overarching organization, things that are common enough for different people that we can actually target them. It’s a bit of a limitless problem that we’ve made huge progress on in the last decade, but still aren’t close to solving.” — Meghan Morrissey

The impact
Today’s immunotherapies are successful in about 25% of patients. A macrophage-based alternative for the 75% not responding to those therapies could potentially boost survival rates for patients with tumor cancers. Importantly, it could prove to be a more targeted and less toxic therapy for all cancer patients.

The work
From remote, rural Colombia to the sunny shores of Santa Barbara, physicianscientist Kenneth S. Kosik delves into the genetic origins of Alzheimer’s disease. His goals: to trace the movement of the disease through the centuries and around the globe, and to find ways to disrupt the formation of the pathological protein tangles and sticky plaques that are the hallmark of the as yet incurable form of dementia that affects millions of people worldwide. His longtime collaboration with renowned Colombian neurologist Francisco Lopera and their fieldwork in Antioquia, Medellín, with the world’s largest genetic cohort of early-onset Alzheimer’s patients, have yielded clinical trials and precious clues in the effort to stop the disease. One promising avenue: A woman in a rural family of thousands predisposed to Alzheimer’s by their 40s, due to a rare genetic mutation, has managed to escape her family’s fate, thanks to a second rare mutation that seems to confer some protection.

The why
“While bench and bedside often seem separated by a wide chasm, the physician-scientist in bridging this divide can discern which of the numerous basic laboratory insights can serve as steps toward treatments for incurable conditions such as Alzheimer’s disease. In so doing, it is an extraordinary privilege to conduct research with the direct participation of those affected, their families, their communities, and with a team of exceptionally talented graduate students and post-doctoral fellows.”
— Kenneth Kosik
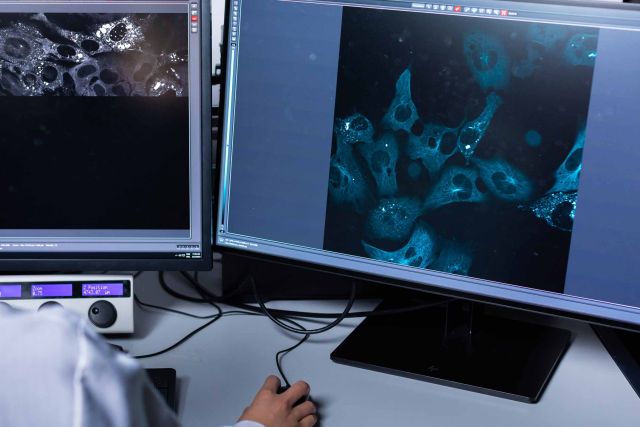
The impact
In the uphill battle that is the search for an Alzheimer’s cure, every finding is an important step toward the day when an Alzheimer’s diagnosis does not automatically consign the patient to a terrible fate.

The work
Few things are as devastating as a diagnosis of age-related macular degeneration, a condition in which retinal cells at the back of the eye break down, causing the loss of the central field of vision. AMD affects more than 200 million people worldwide. But biologist Dennis O. Clegg and collaborators have developed a therapy that has been shown to stop the progress of the disease and even restore some vision in some patients: a “patch” of stem cell-derived retinal pigment epithelium — a layer of darkly colored cells that, when implanted beneath the degrading visual cells, nourishes them and enables their continued function. Early results were promising, with patients with severe AMD experiencing initial improvements and no deleterious effects a year later. As the patch moves toward FDA approval, the team continues to fine-tune it to make it more robust, with the goal of creating an “off-the-shelf” therapy that is easily available to patients everywhere.

The why
“Vision is a precious resource. I am inspired by patients who face the prospect of blindness with no effective treatment available. We have assembled a talented, interdisciplinary team to translate basic research findings to clinical trials with the hope of treating unmet medical needs in eye disease.”
— Dennis Clegg

The impact
A treatment for one of the world’s leading causes of blindness and the preservation and restoration of sight means the improvement and longevity of the independence and the quality of life for the world’s elder population.

The work
Rarely does basic research so directly lead to a clinical impact for people with a progressive, genetic condition as in the case of cell biologist Thomas Weimbs’ work which targets polycystic kidney disease. PKD affects roughly 12 million people worldwide with painful, fluid-filled cysts and inflamed, deteriorating kidneys. While studying animal models with PKD, Weimbs and fellow researcher Jacob Torres were startled to find that in animals that had been fed beta-hydroxybutyrate, a ketone that our bodies make naturally during fasting, the cysts stopped growing and had even shrunk. This led to their realization that PKD can be controlled and even reversed with nutritional interventions, including the popular intermittent fasting and ketogenic diets. Weimbs is currently focusing on clinical trials to determine the long-term efficacy and feasibility of these diets to treat PKD, and, in collaboration with dietitians, he has developed a diet education program and a medical food to help patients control their condition.

The why
“As academic, basic researchers, we are used to publishing our findings and moving on to the next thing. It has been an unexpected and amazing journey to be able to team up with wonderful clinicians and patients with kidney disease, take discoveries straight from the lab and translate them into practice. I would never have thought that our research would lead to the launch of a startup company and the creation of a product that benefits patients. All this happened in record time and my head is still spinning.” — Thomas Weimbs

The impact
Nutritional interventions empower PKD patients with the ability to do something themselves to control an otherwise relentlessly progressive disease, and to preserve their quality of life, rather than waiting for their kidneys to fail.







